 To rank items as equivalent, overlap them. Heat distribution from high temperature to low temperature is a another example of this phenomenorn Part A Rank these systems in order of decreasing entropy. The universe started with low entropy (concentrated in the moment before the "big bang") and the entropy has since been constantly increasing by distributing this energy. The universe has a constant amount of energy as stated in the first law of thermodynamics. Another way of looking at entropy is that the universe is moving toward a broader distribution of energy. The number of molecules, the space available for the molecules, and the energy available to the molecules will impact the number of microstates. 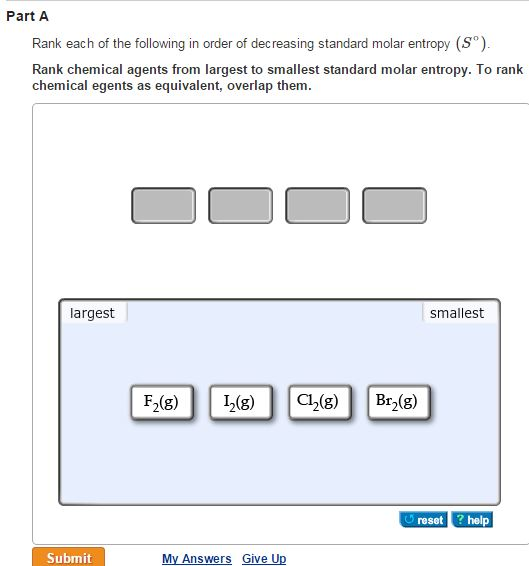
Duing rotational motion, molecules rotate or spin. During vibrational motion, atoms in a molecule move toward and away from one another. 
During translational motion, the entire molecule moves in one direction. 
Molecules may undergo three different types of motion: translational motion, vibrational motion, and rotational motion. The more microstates the system has, the greater its entropy Microstates depend on molecular motion. At the molecular level, entropy can be described in terms of the possible number of different arrangements of particle positions and energies, called microstates. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
